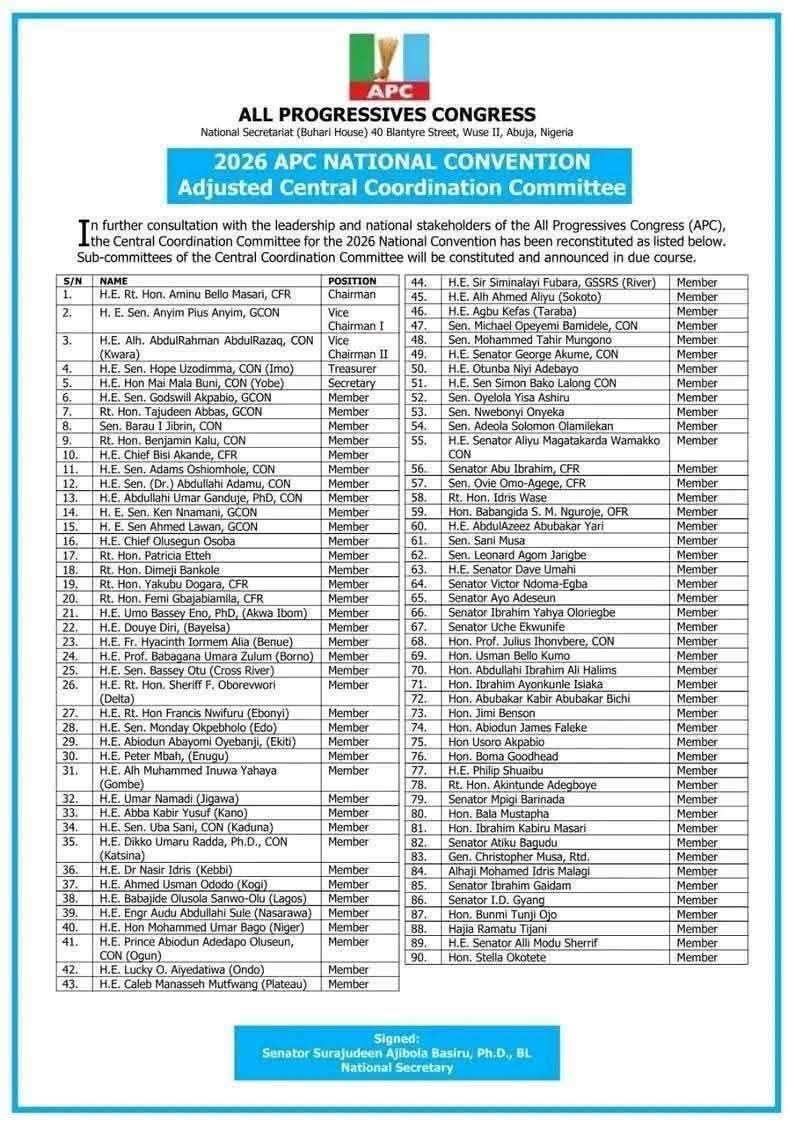
NATIONAL NEWS
NAFDAC Issues Public Alert as Danone Nutricia Recalls Specific Batches of Aptamil, Others

The National Agency for Food and Drug Administration and Control (NAFDAC) has announced a precautionary recall of several batches of Aptamil and Cow & Gate First Infant Milk and Follow-on Milk Formula products manufactured by Danone Nutricia.


The recall follows reports of potential contamination with cereulide, a toxin produced by Bacillus cereus, which can cause foodborne illness characterised by nausea and severe vomiting.
The affected products were manufactured in Ireland and Hungary and distributed across the European Union, the United Kingdom, and other countries.
However, NAFDAC has clarified that Aptamil products specifically produced and registered for the Nigerian market are not affected and remain safe for consumption.
The agency advised consumers, healthcare professionals, caregivers, and distributors to verify product details carefully and report any suspected unregistered or illegally imported products to the nearest NAFDAC office.
Its full statement reads; NAFDAC is notifying the public, healthcare professionals, caregivers, and all relevant stakeholders of the precautionary recall of Aptamil 1 From Birth First Infant Milk, and Follow-on Milk Formula by Danone Nutricia due to potential contamination with cereulide.
Cereulide is a toxin produced by the bacterium, Bacillus cereus, known to cause foodborne illness and may cause symptoms such as nausea and severe vomiting.
Product details:
• Aptamil 1 From Birth First Infant Milk
– Pack size: 800g
– Product Manufacturer: Danone Nutricia Ltd, Ireland
– Expiry Date: 31-10-2026
Other recalled products:
• Aptamil 1 First Infant Milk
– Pack size: 800g
– Best before: All dates from 9 July 2026 to 4 December 2026
– Pack size: 700g
– Best before: All dates from 22 August 2026 to 26 November 2026
• Cow & Gate 1 First Infant Milk
– Pack size: 700g
– Best before: All dates from 31 July 2026 to 31 January 2027
• Cow & Gate 1 First Infant Milk Big Pack
– Pack size: 1.2kg
– Best before: All dates from 11 July 2026 to 20 February 2027
• Cow & Gate 1 First Infant Milk Hungry
– Pack size: 800g
– Best before: All dates from 31 July 2026 to 23 February 2027
• Cow & Gate 1 First Infant Milk
– Pack size: 600g
– Best before: All dates from 12 August 2026 to 16 January 2027
• Cow & Gate Anti-Reflux
– Pack size: 800g
– Best before: All dates from 7 June 2026 to 18 February 2027
• Aptamil 1 First Infant Milk Big Pack
– Pack size: 1.2kg
– Best before: All dates from 14 August 2026 to 25 December 2026
• Aptamil 1 First Infant Milk Hungry
– Pack size: 800g
– Best before: All dates from 4 July 2026 to 24 February 2027
• Aptamil 1 First Infant Milk Pre-Measured Tabs
– Best before: All dates up to and including 17 January 2027
• Aptamil 2 Follow-On Milk
– Pack size: 800g
– Best before: All dates from 18 June 2026 to 4 January 2027
• Aptamil 2 Follow-On Milk
– Pack size: 700g
– Best before: All dates from 5 August 2026 to 20 February 2027
• Aptamil 2 Follow-On Milk Big Pack
– Pack size: 1.2kg
– Best before: All dates from 23 July 2026 to 13 January 2027
• Aptamil 2 Follow-On Milk Pre-Measured Tabs
– Best Before: All dates up to and including 17 January 2027
• Cow & Gate 1 First Infant Milk
– Pack size: 800g
– Best before: All dates from 11 June 2026 to 25 January 2027
The batches in question were manufactured in Ireland and Hungary and distributed across the EU, the UK, and third countries. However, this recall does not impact the Aptamil Infant Formulae produced specifically for Nigeria, which complies with Codex standards and is safe for consumption.
It is important to note that Aptamil Infant Formula and Follow-on/growing-up milks are duly registered for the Nigerian market. Any product without the underlisted 👇 product information and NAFDAC Registration Number is considered illegal within the Nigerian market.
• Aptamil Nutribiotik 1 (Aptamil first infant milk) 800g, 400g
D1-7392
• Aptamil Nutribiotik 2 (Aptamil follow on milk 2) 800g, 400g
D1-7396
•AptaJunior Nutribiotik 3 (Aptamil growing up milk 3) 800g, 400g
D1-7397
Additionally, the company has notified the agency that it has proactively blocked products at the warehouse in Nigeria as a precaution and has begun laboratory analysis. NAFDAC will keep the public updated on developments as necessary regarding these matters.
Consumers are encouraged to report suspected availability of unregistered or illegally imported Aptamil infant formula to the nearest NAFDAC office.
For publication of your news content, articles, videos or any other news worthy materials, please send to newsleverage1@gmail.com. For more enquiry, please call +234-901-067-1763 or whatsapp +234-901-067-1763. To place an advert, please call 09010671763
-

 METRO4 weeks ago
METRO4 weeks agoOnitsha Market Traders Defy Soludo, Insist Sit-at-Home Continues
-

 CRIME4 days ago
CRIME4 days agoArmy Foils Ambush on GOC Convoy by Lakurawa Terrorists, Kills Five Terrorists
-

 Articles2 weeks ago
Articles2 weeks agoObi: Counting the Counters – Abdul Mahmud
-

 NATIONAL NEWS2 weeks ago
NATIONAL NEWS2 weeks agoEl-Rufai Writes NSA Ribadu, Seeks Clarification on Alleged Importation of Poison
-

 NATIONAL NEWS4 weeks ago
NATIONAL NEWS4 weeks agoFull List of Nominees for Nigeria’s Court of Appeal Justices